:max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg) 
This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. Thus, the standard atomic weight of argon, A r°(Ar) =, indicates that atomic-weight values of argon in normal materials are expected to be from 39.792 to 39.963.įor iridium, the standard atomic weight A r°(Ir) = 192.217 ± 0.002 indicates that atomic-weight values of iridium in normal materials are expected to be from 192.215 to 192.219.įor more information on the interpretation of the uncertainty please consult the recent IUPAC Technical Report by Possolo et al.A vertical column in the periodic table. The reported uncertainties of the standard atomic weights are such that the atomic-weight values of normal materials are expected to lie in the given interval with great certitude. (3) our ability to precisely determine the atomic masses of the isotopes.Įlements in the first category are distinguished by an interval standard atomic weight. (2) our ability to determine the isotopic abundances, and (1) well-documented natural variations of isotopic abundances, 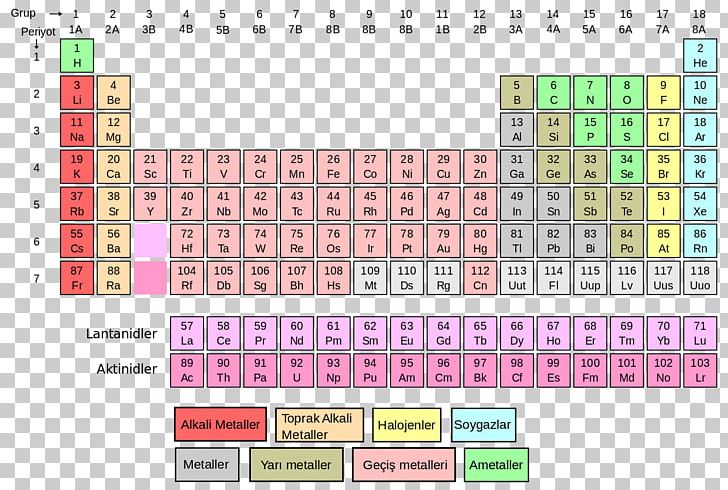
There are three broad groups of elements depending on what is the main cause of the uncertainty of their standard atomic weights: The most recent Standard Atomic Weights are presented in this Table and they are based on the "Atomic Weights 2021" report. R Range in isotopic composition of normal terrestrial material prevents a more precise standard atomic weight being given the tabulated value and uncertainty should be applicable to normal material. Substantial deviations in atomic weight of the element from that given in the table can occur. M Modified isotopic compositions may be found in commercially available material because the material has been subjected to some undisclosed or inadvertent isotopic fractionation. The difference between the atomic weight of the element in such materials and that given in the table may exceed the stated uncertainty. G Geological and biological materials are known in which the element has an isotopic composition outside the limits for normal material. The most recent definitive table of the standard atomic weights. Since 1902, the Commission regularly publishes critical evaluation of atomic weights of elements and below is Standard atomic weights are CIAAW recommended values for atomic weights applicable to all normal materials.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
